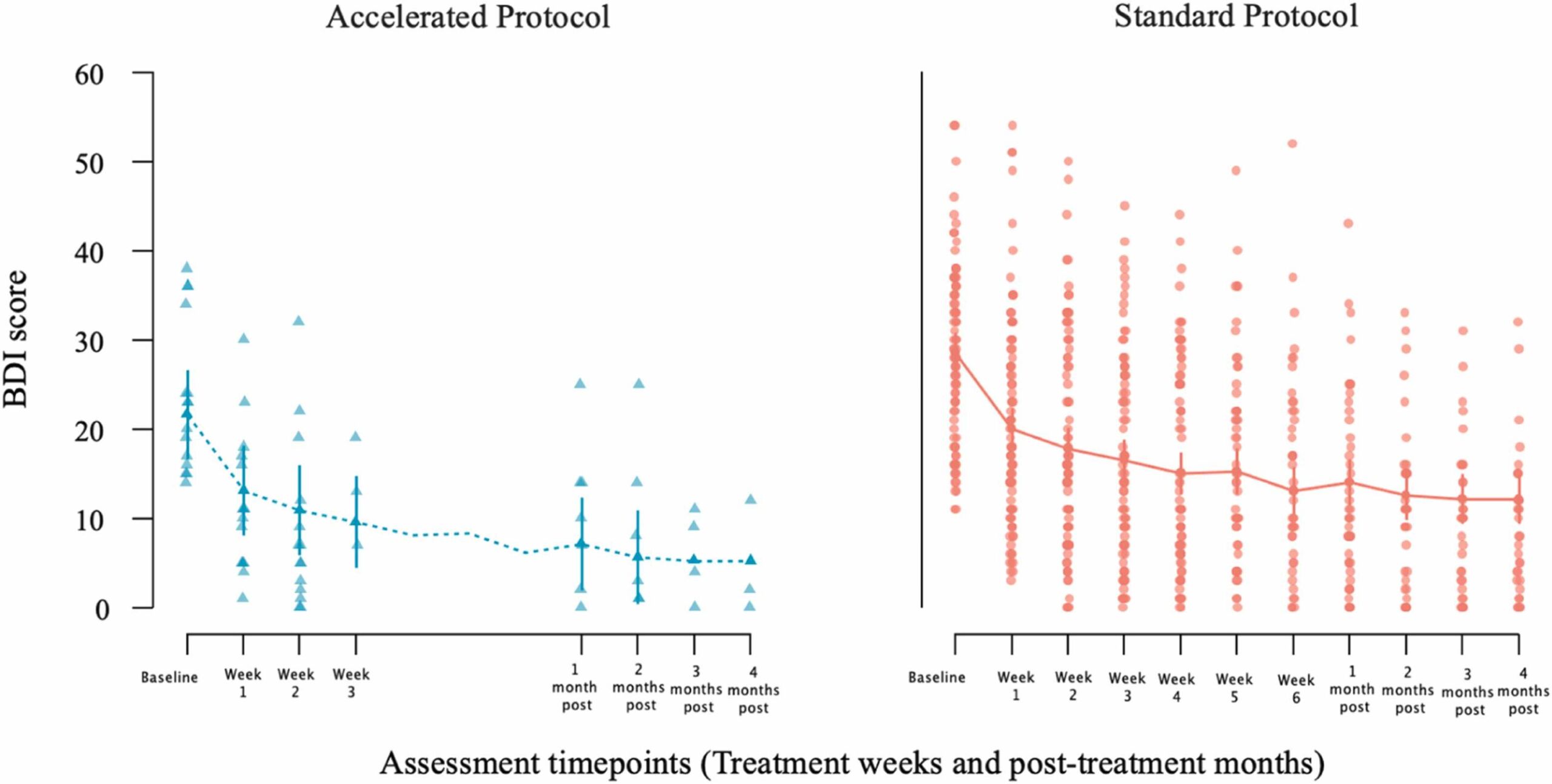
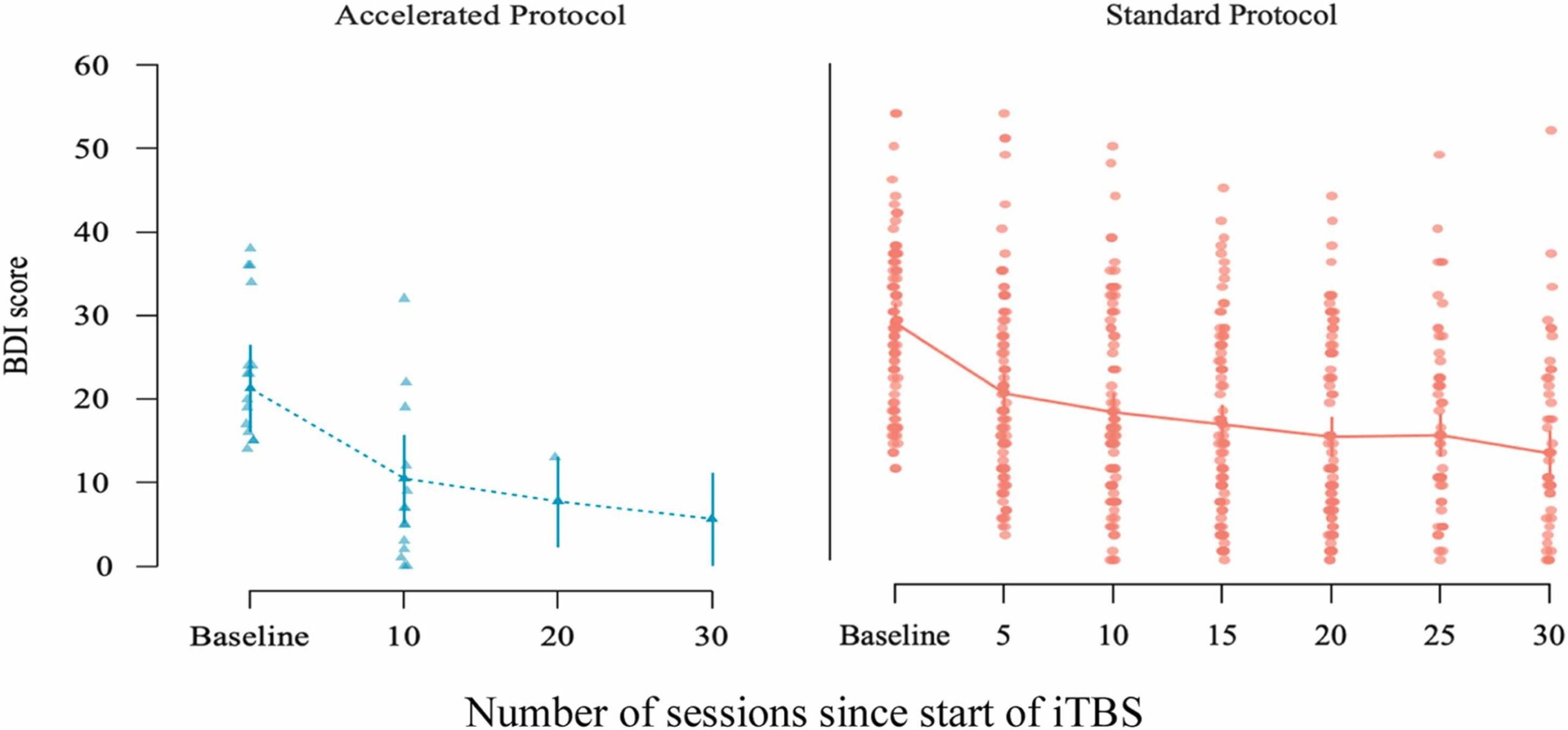
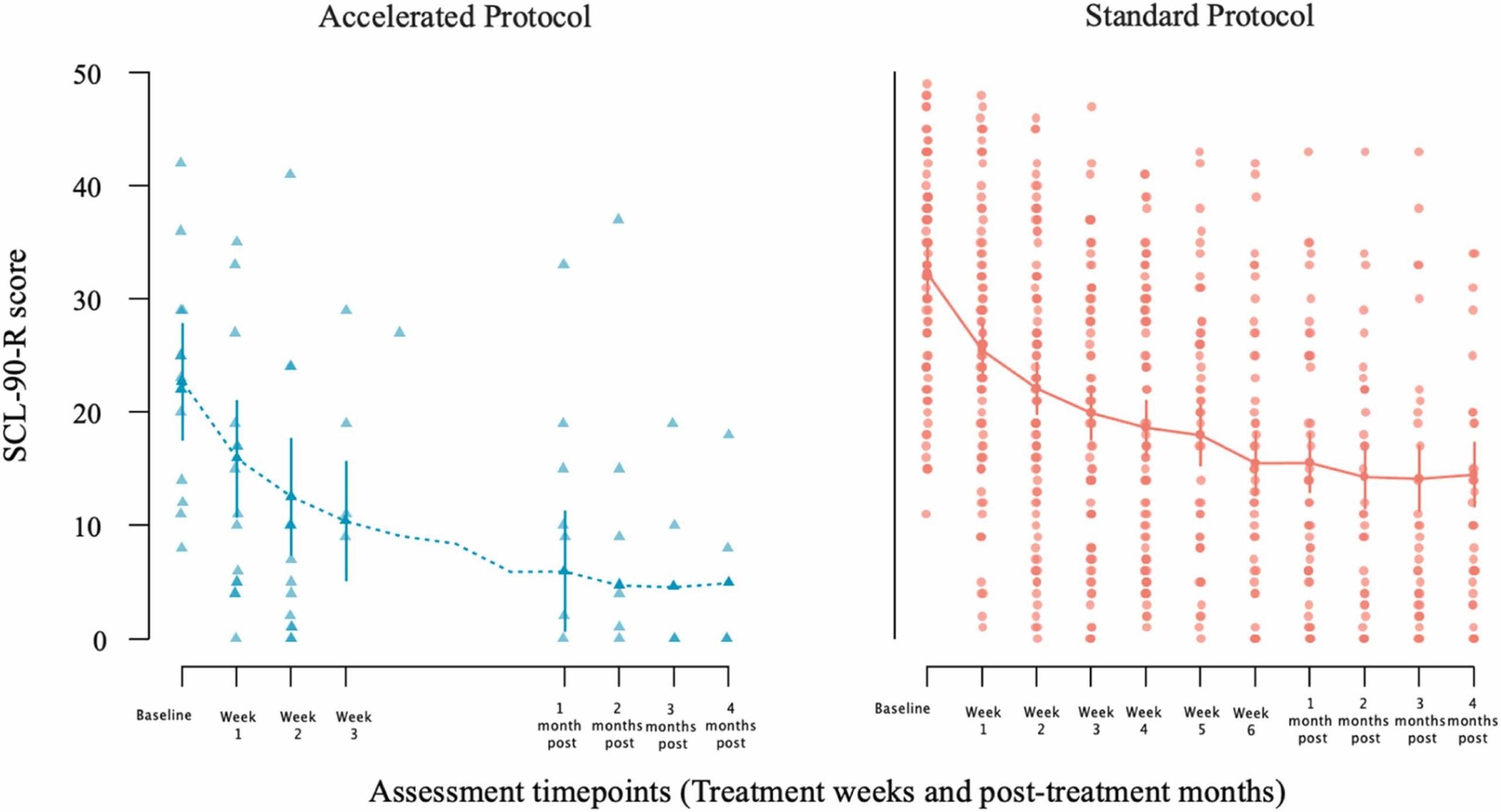
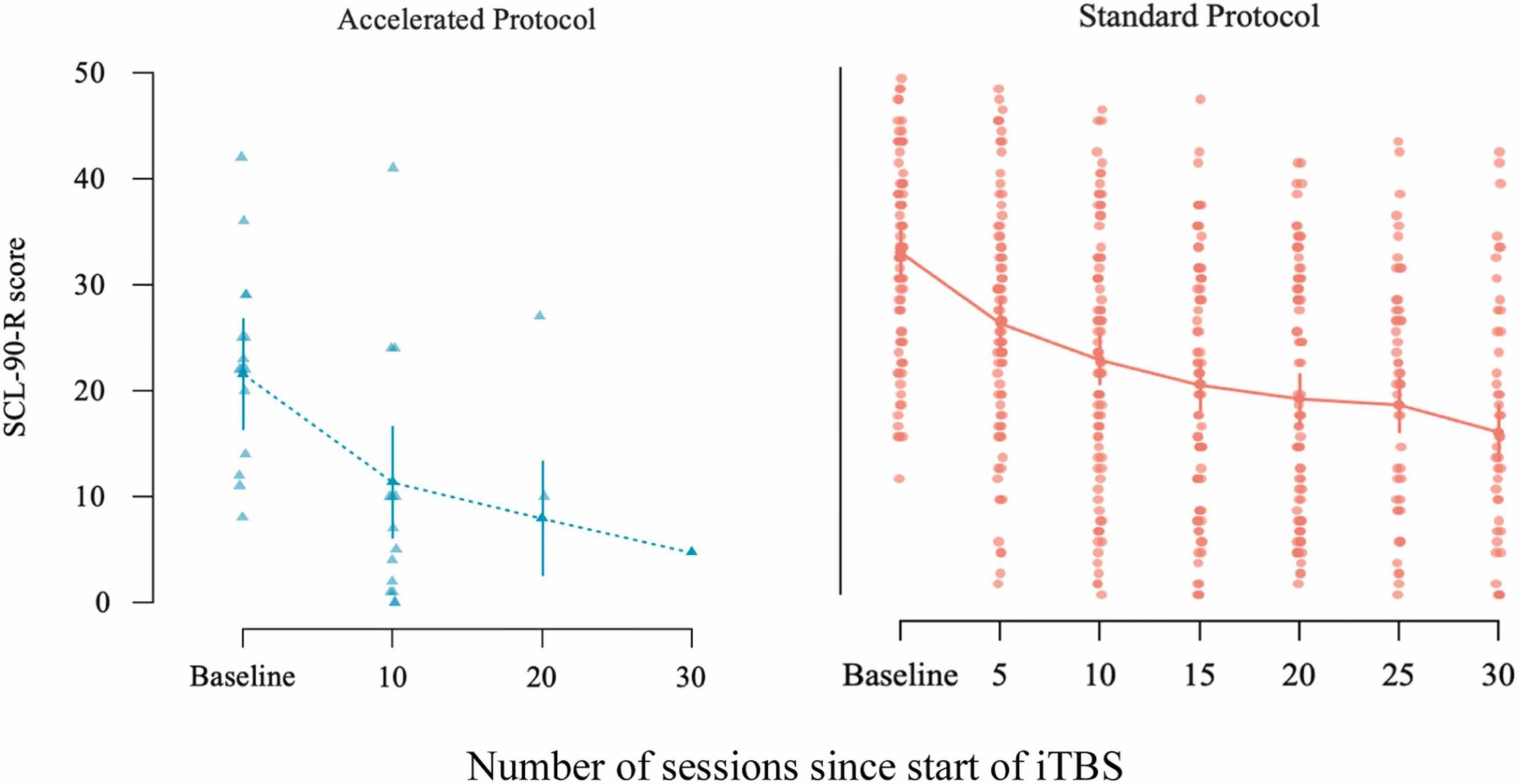
Many patients with depression fail to achieve adequate response or remission with standard treatments, prompting an urgent need to identify factors influencing treatment outcomes. Among these, comorbid personality disorders (PDs) have been identified as a significant contributor to poorer treatment responses.
In this open-label, naturalistic study, we evaluated the feasibility, tolerability, and effectiveness of intermittent theta burst stimulation (iTBS) in a sample of 101 patients with depression and comorbid PD. Eighty-six patients received a standard iTBS protocol (one session per day over several weeks), while fifteen opted for an accelerated protocol involving two to four sessions per day. Symptom severity was assessed using the Beck Depression Inventory (BDI) and the Symptom Checklist-90-R (SCL-90-R), administered at baseline, weekly during treatment, and at four follow-up points (1, 2, 3, and 4 months post-treatment).
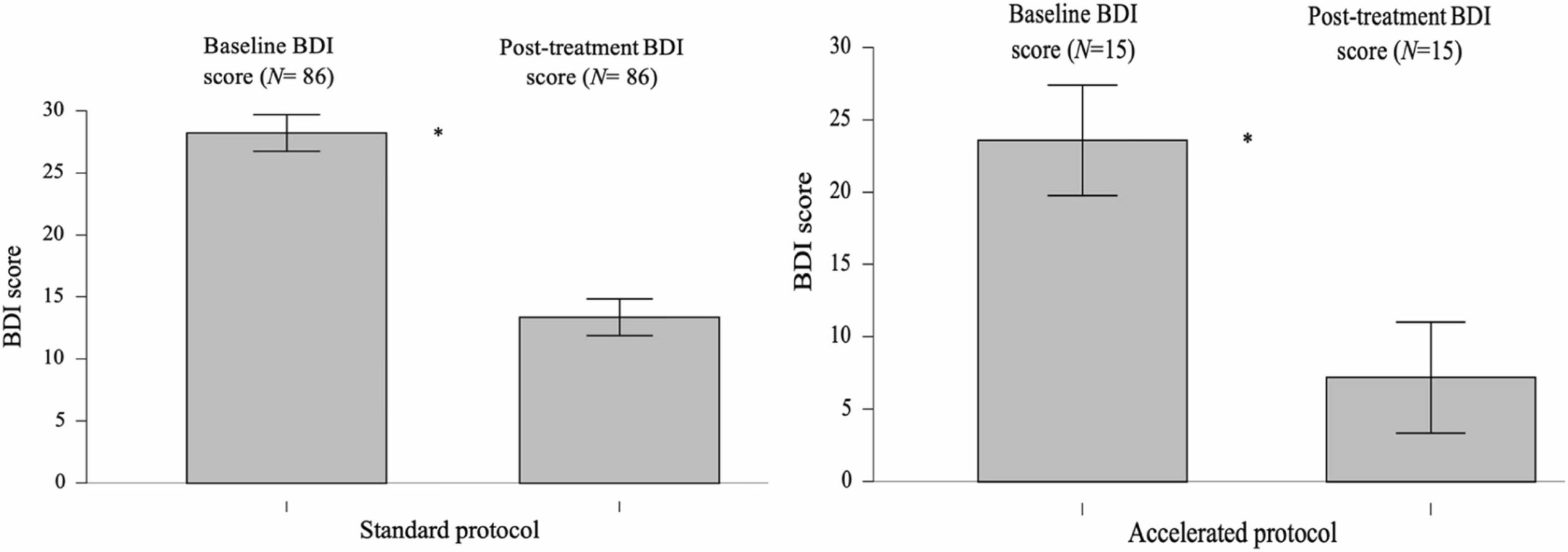
Both treatment protocols led to significant symptom reduction, which was sustained over the follow-up period. Remission rates were 87 % in the accelerated group and 63 % in the standard protocol group. iTBS was well tolerated, with no serious adverse events reported. Notably, patients who were not taking antidepressant medication and those who were younger showed better treatment responses. iTBS appears to be a feasible, safe, and effective treatment option for depressed patients with comorbid PD. The findings suggest the benefits of iTBS treatment and support further exploration of patient characteristics that may predict response to rTMS.
Major Depressive Disorder (MDD) is characterized by diverse physical, psychological, cognitive, and psychomotor disturbances, affecting an estimated 5 % of the global adult population (
American Psychiatric Association, 2000; World Health Organization, 2023). By 2030, MDD is projected to become the leading cause of disability worldwide (
World Health Organization, 2022). The first line of treatment for MDD involves a combination of pharmacological medication and psychotherapy (
Malhi et al., 2020). However, the clinical efficacy of pharmaceutical treatments is highly variable and often accompanied by side effects (
Penn & Tracy, 2012). Alarmingly, approximately 30 % of patients do not show improvement after two cycles of treatment (i.e., the period from when a round of treatment ends until the beginning of the next one) and are subsequently diagnosed with treatment-resistant depression (TRD) (
Lundberg et al., 2023).
Many patients with MDD fail to achieve adequate response or remission with standard treatments (
Kolovos et al., 2017), prompting an urgent need to identify factors influencing treatment outcomes. Among these, comorbid personality disorders (PDs) have been identified as a significant contributor to poorer treatment responses (
Berk et al., 2018; Van & Kool, 2018). Despite the profound impact of PDs on MDD prognosis, they are often overlooked in randomized controlled trials, potentially due to the unstable behaviors that characterize PDs, which increase treatment dropout rates (
Ingenhoven et al., 2010).
PDs are exemplified by enduring behavioural patterns that deviate significantly from cultural norms and are not better explained by other mental disorders or substance use (
American Psychiatric Association, 2000). Globally, PDs have a prevalence of 7.8 % (
Winsper et al., 2020), with affected individuals experiencing heightened morbidity and mortality rates (
Fok et al., 2012). These disorders are often characterised by a distorted sense of one’s self, which may exacerbate depressive symptoms. For instance, avoidant personality disorder (AVDPD) is characterised by low self-esteem, feelings of inferiority and shame, susceptibility to criticism and rejection, lack of energy and social desirability, and inability to feel pleasure (
Young, 2018). Patients with borderline personality disorder (BPD) often experience unstable mood and relationships and feelings of loneliness (
Ceresa et al., 2021).
The management of PDs is complex; while psychotherapy has shown promise, particularly for BPD (
Kramer et al., 2020), the efficacy of pharmacological treatments remains contentious (
Stoffers-Winterling et al., 2021). Interestingly, antidepressants can alleviate depressive symptoms and thereby potentially influencing PD symptoms and diagnosis (
Fava et al., 2002), suggesting the possibility of shared pathophysiological mechanisms between MDD and PDs. The presence of comorbid PDs in MDD poses significant challenges for treatment, as it has been associated with poor prognosis (
Alnæs & Torgersen, 1997; Grilo et al., 2000; Mulder, 2002) and an increased likelihood of TRD (
McIntyre et al., 2023; Young, 2018). The impact of specific PDs on the course of MDD may vary; while BPD is the most common of the PDs co-occurring with MDD (
Ceresa et al., 2021), all PDs can serve as predictors of MDD persistence. Among these, BPD is the most frequent predictor of MDD persistence, followed by schizoid, schizotypal, histrionic, and AVDPD (
Skodol et al., 2011).
Repetitive transcranial magnetic stimulation (rTMS) is well-established as a treatment for MDD due to its capacity to modulate cortical excitability either directly, though excitation or inhibition of targeted cortical areas, or indirectly via broader interconnected neural networks (
Hallett, 2007). However, the presence of comorbid PDs can complicate the treatment of depression (
Newton-Howes et al., 2006). The efficacy of rTMS in patients with both MDD and PD remains underexplored. While some studies have reported clinical improvements in MDD patients with comorbid BPD (
Feffer et al., 2022; Kafas & Umla-Runge, 2024), most of the existing literature focuses specifically on this subtype. Furthermore, clinical trials often exclude participants with psychiatric comorbidities, including PDs, to reduce confounding variables. Although this approach improves internal validity, it significantly limits the generalizability and real-world applicability of findings (
Blanco et al., 2008). In routine clinical practice, patients with MDD frequently present with TRD and/or other comorbidities such as PDs, emphasizing the need for more inclusive research. Further studies are needed to evaluate the effectiveness of TMS in MDD with comorbid PDs.
In light of these challenges, our naturalistic, open-label study primarily aimed to investigate the effects of iTBS on depressive symptoms in MDD patients with comorbid PDs. Participants chose between a standard six-week one session per day iTBS protocol and an accelerated iTBS protocol with multiple sessions, ranging from two to four per day. We also evaluated the safety, tolerability, and practical feasibility of both the standard and accelerated iTBS protocols for treating MDD with comorbid PD.
Our sample consisted exclusively of outpatients who had received an MDD diagnosis with comorbid PD. All patients provided informed consent at the Medical Psychotherapeutic Center (ΙΨΚ) in Thessaloniki, Greece before undergoing treatment. The treatment sessions were conducted at ΙΨΚ between 2018 and 2023. Considering that this was an open-label naturalistic study in a clinical population sample, we were unable to control for variables such as age, sex, medication use and psychotherapy before treatment, and therefore included those as potential covariates in our statistical analyses.
2.2.1 Eligibility criteria
2.2.2 Sample demographics
Each intermittent theta-burst stimulation (iTBS) session consisted of 600 pulses, delivered in bursts of three pulses at 50 Hz, repeated at a frequency of 5 Hz. These bursts were administered in 2-second trains, followed by 8-second inter-train intervals, for a total of 20 trains per session. This protocol is consistent with standard clinical iTBS procedures. All patients (N = 101) were offered a choice between a standard (non-accelerated) and an accelerated iTBS protocol, depending on personal preference and logistical considerations such as work obligations or travel distance. The majority of patients (86 out of 101) chose the standard protocol, receiving one session per weekday over a period of 4–6 weeks, resulting in a total of 20–30 sessions. Fifteen patients opted for the accelerated protocol. Among these, 11 received two sessions per day with a 50-minute break in between, over a period of 2–3 weeks, resulting in a total of 20–30 sessions. Four patients, who were living abroad, completed four sessions per day with 50-minute breaks between each, over a period of one week to 2 weeks resulting in a total of 20–40 sessions.
2.3.3 Beck depression inventory
2.3.4 Symptom checklist 90-R
2.3.5 International personality disorder examination (IPDE)
A linear mixed model (LMM) analysis was used to assess iTBS treatment effects on depression levels over time, measured by BDI scores and the depression subscale of the SCL-90-R. Time (pre-treatment vs. post-treatment) was a within-subject factor, while Protocol Type (accelerated vs. standard) was a between-subject factor. Covariates included: sex, age, total number of classes of medications, number of benzodiazepines, adjunct psychotherapy, TMS dosage (i.e., stimulation intensity), and number of sessions.
As our data lacked independency, in view of our repeated measures design, and due to the fact that the two protocol groups were unbalanced, we chose LMM as the preferred analysis method. LMMs can model unequal sample sizes and are also appropriate for continuous outcomes, while they allow for random effects, accounting for unexplained variance within our patient sample. Data was normally distributed as skewness and kurtosis for both BDI (skewness =.59; kurtosis = −.29). and SCL-90-R (SCL-90-R skewness =.52; kurtosis =.15) were within normal ranges. Q-Q plots indicated that both assumptions of normality and linearity were met for BDI and SCL-90-R.
PD effects were analyzed through four models examining PD clusters and individual PD diagnoses. A backwards selection process simplified the initial comprehensive model, retaining relevant fixed factors; PD clusters (centered); and interaction terms involving Time, Protocol, and PDs. Individual PD models included centered PD diagnoses and interaction terms up to four-way interactions, with BPD and AVDPD prioritized due to prevalence. Antisocial PD was excluded due to insufficient observations.
Post-hoc Bonferroni-corrected pairwise comparisons identified time points of significant changes between protocols (p < .05). We used a Likelihood Ratio (RT) test and chose the Bayesian Information Criterion (BIC) for selecting the best fitting model. A random intercept of person was fitted to the data by default to account for individual variation in baseline scores, but a random slope for time was removed due to too few observations.
In addition, we conducted post hoc independent t-tests to explore pre-, and post-treatment changes in patients’ depression levels, as measured by the BDI, between the two protocol groups (i.e., accelerated vs. standard). Paired student’s t-tests were conducted for investigating pre-, and post- treatment changes in BDI scores for each PD cluster and each PD separately. A correlation analysis and a one-way ANOVA examined practical feasibility as well as the association between TMS dosage and medication prescription. All analyses were conducted using JASP v.0.18.3 with a standard covariance matrix for random effects.
3.1 Safety and tolerability
3.2 Practical feasibility
3.3 Response and remission rates
Fig. 3 Change in patients’ SCL-90-R scores from baseline to end of treatment [week 3 for the accelerated group (left panel); week 6 for the standard group (right panel)] and from baseline to post-treatment follow-up month (first through fourth post months) assessments after the end of each patient treatment. Dots represent individual patient scores; lines indicate group means with standard error bars.
3.4 BDI outcomes: effects of iTBS treatment
3.4.1 Model 1: PD clusters
3.4.2 Model 2: specific PD diagnoses
3.5 SCL-90-R outcomes: effects of iTBS treatment
3.5.1 Model 1: PD clusters
3.5.2 Model 2: specific PD diagnoses
3.6 Impact of personality disorder traits on treatment efficacy
4.1 Limitations and future research
Our open-label naturalistic study assessed the feasibility, tolerability, and effectiveness of a repetitive transcranial magnetic stimulation in 101 patients with depression and comorbid PD. The findings indicate that both the standard and accelerated protocols are feasible, well-tolerated and clinically effective in this population. Larger studies are needed to confirm the effectiveness of the accelerated protocol. Given that iTBS requires less time than conventional rTMS protocols, it offers greater flexibility and reduced time burden for both patients and clinicians. These promising results underscore the importance of follow-up research using randomized controlled trial designs.
CRediT authorship contribution statement
Theodoros Koutsomitros: Writing – review & editing, Writing – original draft, Visualization, Validation, Supervision, Software, Resources, Project administration, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Panagiota Koutsimani: Writing – review & editing, Writing – original draft, Visualization, Formal analysis. Teresa Schuhmann: Writing – review & editing, Writing – original draft, Visualization, Validation, Supervision, Software, Project administration, Methodology, Investigation, Formal analysis, Data curation. Alexander T. Sack: Writing – review & editing, Writing – original draft, Visualization, Validation, Supervision, Software, Resources, Project administration, Methodology, Investigation, Formal analysis, Conceptualization.
This research did not receive external funding.
Declaration of Competing Interest
We would like to thank all the psychologists and psychiatrists at the Medical Psychotherapeutic Centre in Thessaloniki, Greece, who referred patients for this study.
Appendix A Supplementary material (1)